
Die Patient Voice Database ermöglicht es Ihnen, bessere Behandlungen zu gestalten, indem sie Sie sicher und stets zu Ihren Bedingungen mit der Forschung verbindet.
Sie werden gebeten, einige Fragen unserer künstlichen Intelligenz zu beantworten, basierend auf den Tausenden von Studien, die derzeit rekrutieren.

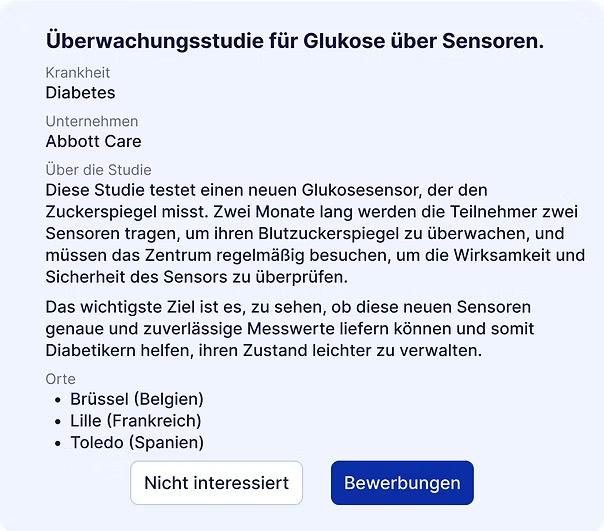
Basierend auf Ihren Antworten generiert Curewiki eine Liste von Studien, die Ihrem Profil und Ihren Präferenzen entsprechen.
Sie entscheiden, ob Sie sich freiwillig melden und mit dem Hauptprüfer in Kontakt treten möchten.
Die Registrierung verpflichtet Sie zu nichts weiter als zum Erhalt von Studienvorschlägen.

Beantworten Sie ein paar Fragen — keine medizinischen Unterlagen erforderlich. Es ist privat und sicher.

Wir zeigen Ihnen nur die Möglichkeiten, die Ihren Bedürfnissen entsprechen, und niemand kontaktiert Sie ohne Zustimmung.

Interessiert? Verbinden Sie sich. Noch nicht bereit? Ignorieren Sie es. Es liegt immer an Ihnen.
"Jeder hat das Recht, an medizinischer Forschung teilzunehmen. Deshalb öffnen wir den Zugang zu allen klinischen Studien, die in Europa rekrutieren. Darüber hinaus maximiert unsere Unabhängigkeit Ihre Chancen, die richtige Studie für Ihre Bedürfnisse zu finden."
Jean-Sébastien, Gründer von Curewiki
Curewiki ist ein europäisches Startup (12 Personen in den Niederlanden, Belgien und Spanien), das 2022 gegründet wurde und mit öffentlichen und privaten Mitteln finanziert wird.

Als Überlebender von Kinderkrebs möchte Jean-Sebastien, dass jeder eine Behandlung erhält, die ihm hilft zu heilen. Er leitet das Unternehmen.

Ein ehemaliger Arzt, der mit der Schwierigkeit zu kämpfen hatte, klinische Studien für Patienten und Bekannte zu finden, gründete Edwin ein medizinisches Forschungsunternehmen, bevor er zu Curewiki kam. Er ist für die Geschäftsbeziehungen zuständig.

Mit 15 Jahren Erfahrung in Fintech, Health Tech und Einzelhandel leitet Mieke Strategien, die Patienten mit klinischen Studien verbinden, mit Innovation und messbarer Wirkung.

Mit über 10 Jahren Erfahrung in Technologie und digitalem Geschäft treibt Olga die Marktexpansion voran und baut wirkungsvolle Partnerschaften für Wachstum auf.

HÖREN SIE NICHT AUF ZU GLAUBEN.
Haben Sie Fragen? Schauen Sie nach, ob wir bereits eine Antwort für Sie haben.